Website owner: James Miller
Gram-equivalent weight
Def. Gram-equivalent weight. The mass of a substance, in grams, that combines with or is chemically equivalent to, one gram-atomic weight (1.008 grams) of hydrogen: the atomic or molecular weight divided by the valence.
Syn. Equivalent weight, gram-equivalent
Gram-equivalent weights for various cases:
1. Complex ion. The gram-equivalent weight of a complex ion is equal to the formula weight of the ion divided by its valence.
Examples. The gram-equivalent weight of the OH - ion is 16.000 + 1.008 = 17.008 g and of the Ca++ ion is 40.08/2 = 20.04 g.
2. Acid. The gram-equivalent weight of an acid is that weight of the acid that contains 1 gram-equivalent weight (1.008 g) of replaceable hydrogen.
Examples. 1) The gram-equivalent weight of HCl is one mole of HCl (1.008 + 35.453 = 36.461 g). 2) The gram-equivalent weight of H2SO4 is usually ½ mole since both hydrogens are replaceable in most reactions of sulfuric acid. 3) The gram-equivalent weight of H3PO4 may be 1 mole, ½ mole, or ⅓ mole, depending on whether 1, 2, or 3 hydrogen atoms per molecule are replaced in a particular solution. 4) The gram-equivalent weight of H3BO3 is always one mole, since only one hydrogen is replaceable.
There are no simple rules for predicting how many hydrogens of an acid will be replaced in a given reaction.
3. Base. The gram-equivalent weight of a base is that weight of the base that contains 1 gram-equivalent weight (17.008 g) of replaceable OH - .
Examples. The gram-equivalent weight of NaOH is 1 mole (i.e. 1 gram-formula weight) , that of Mg(OH)2 is ½ mole, and of Al(OH)3 is ⅓ mole.
4. Simple normal salt. The gram-equivalent weight of a simple normal salt that does not act as an oxidizing or reducing agent is that weight of the salt which contains 1 gram-equivalent weight of the metal or acid radical.
Examples. The gram-equivalent weight of NaCl is 1 mole (i.e. 1 gram-formula weight), that of NH4Cl is1 mole, that of CaCl2 is ½ mole, that of AlF3 is ⅓ mole, and that of Mg3(PO4)•4H2O is 1/6 mole.
In general, the gram-equivalent weight of a salt in which a change of valence does not occur during the reaction is equal to its gram-formula weight divided by the total charge of all positive ions (or of all negative ions).
5. Acid and complex salts. The gram-equivalent weights of acid and complex salts depend on the metal, the unreplaced acid hydrogen, or the acid radical, with reference to which the equivalent is to be calculated.
Example. The gram-equivalent weight of the acid salt NaH2PO4 would be 1 mole (i.e. 1 gram-formula weight), ½ mole, or ⅓ mole, depending on whether the salt is used for its Na, total H or PO4 content.
Equivalent weight of an oxidizing or reducing agent. The equivalent weight of an oxidizing or reducing agent for a particular reaction is equal to its formula weight divided by the total number of electrons gained or lost when the reaction occurs (i.e. by the total change in valence).
Example. Q. In a given reaction MnO2, in HCl, solution, is reduced to MnCl2. What is the equivalent weight of MnO2 in this reaction?
A. In the reaction
MnO2 + 2 HCl →MnCl2 + H2O
Mn has a valence of +4 in MnO2 and a valence of +2 in MnCl2. The change in its valence is thus 2 and
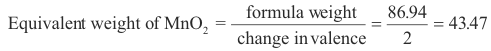
Note that Mn is the only element in MnO2 whose valence changes during the reaction (by convention the valence of oxygen is always -2 except in peroxides). Thus the change in valence of Mn represents the total change in valence of the atoms comprising MnO2.
A given oxidizing or reducing agent may have more than one equivalent weight, depending on the reaction for which it is used.
References
Schaum, Beckman, Rosenberg. College Chemistry. (Schaum)
Jesus Christ and His Teachings
Way of enlightenment, wisdom, and understanding
America, a corrupt, depraved, shameless country
On integrity and the lack of it
The test of a person's Christianity is what he is
Ninety five percent of the problems that most people have come from personal foolishness
Liberalism, socialism and the modern welfare state
The desire to harm, a motivation for conduct
On Self-sufficient Country Living, Homesteading
Topically Arranged Proverbs, Precepts, Quotations. Common Sayings. Poor Richard's Almanac.
Theory on the Formation of Character
People are like radio tuners --- they pick out and listen to one wavelength and ignore the rest
Cause of Character Traits --- According to Aristotle
We are what we eat --- living under the discipline of a diet
Avoiding problems and trouble in life
Role of habit in formation of character
Personal attributes of the true Christian
What determines a person's character?
Love of God and love of virtue are closely united
Intellectual disparities among people and the power in good habits
Tools of Satan. Tactics and Tricks used by the Devil.
The Natural Way -- The Unnatural Way
Wisdom, Reason and Virtue are closely related
Knowledge is one thing, wisdom is another
My views on Christianity in America
The most important thing in life is understanding
We are all examples --- for good or for bad
Television --- spiritual poison
The Prime Mover that decides "What We Are"
Where do our outlooks, attitudes and values come from?
Sin is serious business. The punishment for it is real. Hell is real.
Self-imposed discipline and regimentation
Achieving happiness in life --- a matter of the right strategies
Self-control, self-restraint, self-discipline basic to so much in life