Website owner: James Miller
Radioactivity. Alpha and beta particles. Gamma rays. Wilson cloud chamber. Geiger counter. Decay chain of radium and uranium. Transmutation of an element. Particle accelerators. Van de Graaff generator. Cyclotron. Synchrotron. Nuclear fission. Chain reaction. Discovery of the electron, proton, and neutron.
Today’s physicists fire high speed particles at the nuclei of atoms with the idea of smashing them and examining the nuclear debris for clues to the basic structure of matter. It all started with the discovery of radioactivity.
Radioactivity. In 1857 a Frenchman, Niépce de Saint-Victor, noted that certain uranium salts could expose photographic plates, even in complete darkness. In 1896 another Frenchman, Henri Becquerel, investigated the phenomenon with experiments that showed that compounds of uranium apparently gave off a kind of radiation that could pass through thick layers of black paper to expose photographic plates, even in a dark drawer. Various experimenters became interested in the phenomenon and started investigating it, including Becquerel, Pierre and Marie Curie, Paul Villard and Ernest Rutherford.
Rutherford showed that the emanations given off by uranium sulfate were capable of ionizing the air in the space between the charged metal plates of an ionization chamber. The current registered by a galvanometer in series with the circuit was taken as a measure of the ionizing ability and viewed as a measure of the “activity” of the compound.
Mme. Curie discovered that pitchblende, a mineral containing uranium, was four times as active as pure uranium and concluded that the pitchblende must contain another substance more active than uranium itself. She patiently worked over several tons of pitchblende and succeeded in isolating a few milligrams of the bromide of an element that was 1,000,000 times as active as uranium. She called the new element radium.
This radiation produced by compounds of uranium, radium and certain other substances (thorium, for example) was shown by researchers to be caused by the spontaneous breakdown of atomic nuclei in a phenomenon termed radioactivity. The radiation was gradually discovered to be of not a single type, but to be one or more of the following three types of emission:
● alpha particle emission where alpha particles consist of two protons and two neutrons i.e. the nucleus of a helium atom
● beta particle emission where beta particles are electrons traveling at tremendous speeds, some near the speed of light
● gamma ray emission where gamma rays are electromagnetic rays of very high energy and short wavelength
Some substances may emit alpha particles, some beta particles. Some of these emissions may be accompanied by gamma rays.
Alpha particles. Alpha particles have a velocity of about 10,000 miles/sec. Their penetrating power is not very great. They can be stopped by a thin piece of aluminum foil or a thin sheet of paper. Alpha particles are very efficient in ionizing gas molecules in the air. They have an electrical charge of +2.
Beta particles. Beta particles have velocities ranging from 60,000 to 180,000 miles/sec and have much greater penetrating power than alpha particles.

Gamma rays. Gamma rays appear to be high energy X-rays. They are much more penetrating than either alpha or beta particles.
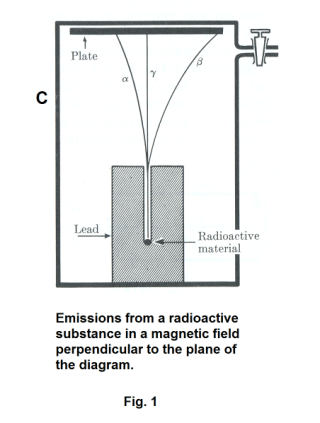
The fact that the radiation does consist of three different types of emission can be shown using the apparatus shown in Fig. 1. A small piece of radioactive material is placed at the bottom of a long groove in a lead block which sits on the bottom of an air tight container C. A photographic plate is placed at a distance above the block and the container is highly evacuated. A strong magnetic field is applied perpendicular to the plane of the diagram, directed into the page. On developing the plate, three spots are found, one in the direct line of the groove, one to the left side of the line and one on the other side. The heavy alpha particles are deflected slightly to the left, the beta particles are deflected substantially more to the right, and the gamma rays are not deflected at all.
Def. Radioactivity. The spontaneous, uncontrollable breakdown of the nucleus of an atom with the emission of particles and electromagnetic rays.
Def. Nuclear radiation. The particles or rays that are given off from a radioactive element, such as uranium, as it decays i.e. the alpha, beta and gamma radiation that may be emitted.
Methods of detecting and measuring nuclear radiation. Alpha and beta particles
ionize substances through which they pass, the degree of ionization being proportional to the intensity of the radiation. This fact is used in the different methods of detecting and measuring the radiation. The following methods can be used to detect or measure nuclear radiation:
1. The electroscope. Because an electroscope becomes discharged from ions in the surrounding air, the rate at which it becomes discharged can be used as a measure of the amount of radiation in the air.
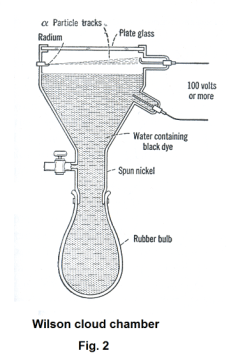
2. The Wilson cloud chamber. The Wilson cloud chamber can be used to detect particle trajectories. A diagram of the chamber is shown in Fig. 2. The lower part of the inverted cone / rubber bulb assembly is filled with water that is dyed black. A small compartment in the upper left side contains a small sample of the radioactive substance to be studied. A voltage of 100 volts or more is maintained across the air space in the upper compartment, which is artificially illuminated.

When the rubber bulb is squeezed, the air in the chamber is compressed. When it is released, the air expands and tracks of the alpha or beta particles can be seen through the glass window at the top, against the black background. See Fig. 3. The particle tracks become visible because they ionize the gas molecules in the air and particles of fog condense on the ionized particles.
3. The bubble chamber. The bubble chamber is used in a way similar to the cloud chamber to detect and record the tracks of electrically charged particles. It is similar to the cloud chamber in basic operating principle. A large cylinder is filled with a transparent liquid (most often liquid hydrogen) heated to just below its boiling point. As particles enter the chamber, a piston suddenly decreases its pressure, and the liquid enters into a superheated state. Charged particles create an ionization track, around which the liquid vaporizes, forming microscopic bubbles which grow until they are large enough to be seen and photographed. The entire chamber is subject to a constant magnetic field, which causes the charged particles to travel in helical paths. Cameras photograph events.
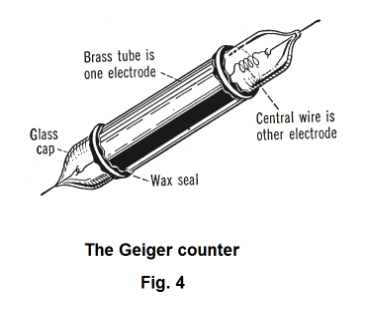
4. The Geiger counter. The Geiger counter is used to detect and measure radiation in the form of alpha, beta or gamma rays. See Fig. 4. It consists of a metal cylinder, which acts as one electrode, surrounding a thin wire, which is the other electrode. By using glass caps, the two electrodes are separated from one another. The cylinder is filled with an inert gas such as helium, neon, or argon at low pressure and a difference of potential is maintained between the electrodes that is just slightly short of that required to produce a discharge. An alpha, beta or gamma ray that enters the counter ionizes the gas molecules and causes a very brief current that is amplified and registered as a click in a loudspeaker.
5. The wire chamber. A wire chamber is a chamber with many parallel wires, arranged as a grid and put on high voltage, with the metal casing being on ground potential. An incoming charged particle leaves a trace of ions and electrons, which drift toward the case or the nearest wire. By noting the wires which had a pulse of current, one can trace the particle's path.
Properties of radium. Radium has the following properties:
1. It affects a photographic plate, even through opaque substances such as paper, wood and thin sheets of metal.
2. Some radium salts can produce fluorescence.
3. It is active chemically. It decomposes water, changes oxygen to ozone, and gives glass a purple color.
4. It produces important physiological changes. It can destroy the germinating power of seeds, kill bacteria, and produce terrible burns that take a long time to heal.
5. The salts of radium glow in the dark, producing a pale phosphorescence.
6. It gives off about 120 calories of heat per gram per hour.
7. It is rare and expensive.
The disintegration of a radium atom. The nuclei of radium and other radioactive
substances are continually disintegrating. The nuclear radiation that they emit are products of this atomic disintegration.
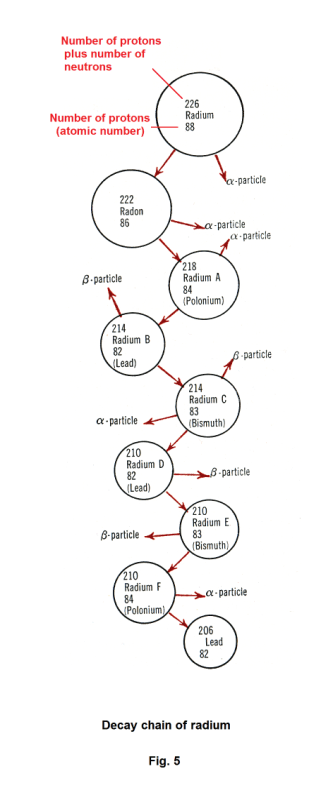
The disintegration of radium occurs through a series of steps in the following decay chain. See Fig. 5.
Step 1. The radium atom emits an alpha particle and disintegrates into Radon by the nuclear equation
88Ra226 → 86Rn222 + 2He4
where the subscript is the number of protons in the nucleus (the atomic number) and the superscript is the number of protons plus the number of neutrons in the nucleus (called the mass number).
Step 2. The radon atom emits an alpha particle and disintegrates into Polonium (Radium A) by the equation
86Rn222 → 84Po218 + 2He4
Step 3. The Polonium atom emits an alpha particle and disintegrates into an isotope of lead (Radium B).
Step 4. Radium B emits a beta particle and disintegrates into an isotope of Bismuth (Radium C) by the equation
82Pb214 → 83Bi214 + e
Note that the mass number 214 has remained the same but a proton has been gained, going from 82 protons in Pb to 83 protons in Bi. What has happened is that in the emission of a beta particle (an electron), a neutron has become a proton! A very remarkable phenomenon! A neutron would appear to consist of a proton and an electron.
Rule. When a beta particle is emitted, a neutron becomes a proton.
Step 5. Radium C emits an alpha particle and a beta particle and disintegrates into an isotope of lead (Radium D) by the equation
83Bi214 → 82Pb210 + 2He4 + e
As can be seen from Fig. 5, this decay chain continues until lead, 82Pb206, is reached.
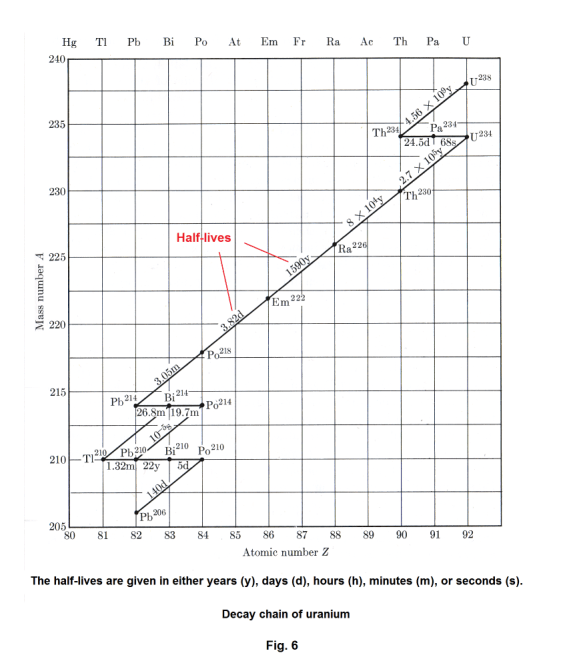
Uranium decay chain. The decay chain of uranium is shown in Fig. 6. The atomic number is plotted along the x axis and the mass number (number of protons plus number of neutrons) is plotted along the y axis. The half-lives are given in either years (y), days (d), hours (h), minutes (m), or seconds (s).
Def. Transmutation of an element. The changing of one element into another through the alteration of its nuclear structure.
First successful human-produced transmutation of an element. So far we have considered transmutations of elements that occur naturally through the process of radioactive decay. Around 1919, Rutherford performed several experiments to find out if it was possible to artificially alter the nucleus of an atom to produce a different element. In one experiment he bombarded nitrogen with alpha particles from radium and found that hydrogen and an isotope of oxygen had been produced according to
2He4 + 7N14 → 8O17 + 1H1
This is the first time man had successfully changed one element into another.
Particle accelerators, atom smashers. Alpha particles from radium are not very effective in bringing about changes in the nucleus of an atom. They have little penetrating power. Scientists consequently sought a new source of particles with which to bombard nuclei. One of the devices constructed was the Van de Graaff generator.
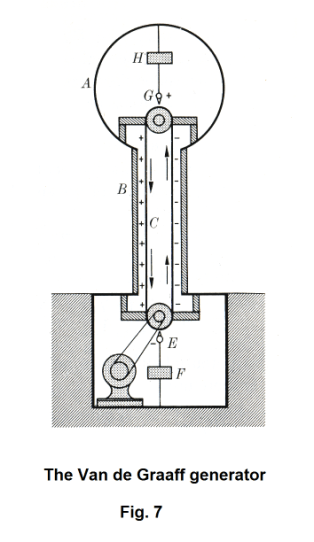
Van de Graaff generator. Fig. 7 shows a schematic diagram of a Van de Graaff generator. The main principle behind the Van de Graaff generator is that if a charged conductor is brought into internal contact with a second hollow conductor, all of its charge transfers to the hollow conductor no matter how high the potential of the later may be. Thus, if it weren’t for insulation difficulties, the charge, and hence also the potential, of a hollow conductor could be raised to any desired value by successively adding charges to it. In practice, because of charge leakage to the supporting structure and the surrounding air, the maximum potential is limited.
Referring to Fig. 7, A is a hollow spherical conductor supported on an insulating hollow column B. Electrons are put on a moving belt C at point E which is maintained at a negative potential tens of thousands of volts relative to ground. They travel to the top and are picked off by collector G, from which they are transferred to the spherical terminal A.
To keep the high charge on a Van de Graaff generator from leaking off into the air, they are sometimes built within a large pear-shaped shell that is kept pressurized with dry air.
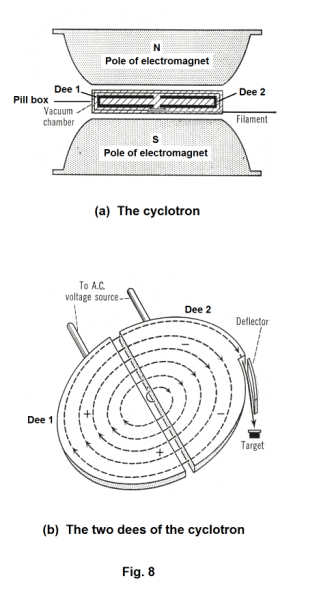
The cyclotron. The cyclotron, a particle accelerator that was invented in 1931 by E. O. Lawrence of the University of California, is used to accelerate massive particles such as protons, deuterons, alpha particles, etc. It consists of a large cylindrical box, shaped like a pill box, placed between the poles of a huge electromagnet. See Fig. 8 (a). Inside this pill box are two hollow D-shaped electrodes called dees. See Fig. 8 (b). The pill box / dee assembly is exhausted to create a very high vacuum inside. The particles that are to be accelerated are fed into the center of the box. As shown in Fig. 8 (b) the charged particles accelerate outwards from the center of the box along a spiral path. The particles are held to a spiral trajectory by the static magnetic field of the two magnets and accelerated by a rapidly alternating (radio frequency) electric field existing in the gap between the dees. This alternating electric field between the two dees is created by connecting the two dees to an alternating AC voltage source, one dee to one side of the line, the other dee to the other. Thus, on one half cycle Dee 1 is positive and Dee 2 is
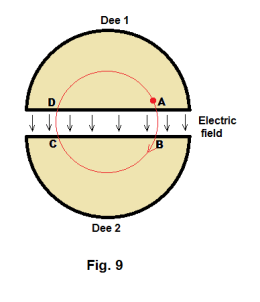
negative; and on the next half cycle, Dee 1 is negative and Dee 2 is positive. This alternating electric field changes in direction in sync with the moving particle in such a way that when the particle crosses a gap between the dees the field is always of the right direction to give it a push and increase its velocity (always of the direction to aid it, boost it). Thus when the particle moves across the gap from A to B in Fig. 9, the electric field is directed from A to B. When the particle reaches point C and moves across the gap from C to D, the electric field has changed direction and is directed from C to D.
In understanding how the cyclotron works, one must keep a couple of facts in mind:
1. A charged particle moving in a plane perpendicular to a magnetic field moves in a circle provided no forces other than the field act on it.
2. No electric field exists inside the dees since an electric field cannot exist inside a hollow conductor.
For the case of a charged particle moving in a plane perpendicular to a magnetic field, the radius r of the circle in which the particle moves is given by the formula
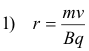
where v is the velocity of the particle, m is the mass of the particle, B is the strength of the magnetic field, and q is the charge of the particle. The angular velocity of the particle is given by ω = v/r. Thus, from 1), we have
![]()
and we see from this formula that the angular velocity is independent of the speed of the particle and of the radius of the circle in which it travels. Thus, if the electric field reverses at regular intervals, each equal to the time it takes the particle to make a half revolution, the electric field in the gap will always be of the right direction to accelerate the particle when it crosses the gap.
The energy of the particles accelerated in a cyclotron may reach 15,000,000 electron-volts. This is the energy an electron would have if it were accelerated across a potential difference of 15,000,000 volts.
The cyclotron operates well only with relatively massive particles such as protons or deuterons. Problems are encountered when trying to accelerate electrons because, to reach the same level of energy that heavier particles can attain, electrons must be accelerated to much higher velocities, and when electrons are accelerated to velocities close to the speed of light, their mass increases causing them to get out of step with the phase of the electric field when they arrive at the gaps.
The synchrotron. The synchrotron also works on the principle of accelerating particles by causing them to move in a circular path with increasing velocity. However, with the synchrotron both the oscillating voltage and the magnetic field are variable and controllable. This capability allows the synchrotron to adjust the electric field synchronisation for increasing mass from high velocities and allow much higher velocities.
The synchrotron can impart energies of several billion electron volts to the protons that they accelerate.
Discovery of the neutron. Through the 1920s, physicists had generally accepted an incorrect model of the atomic nucleus, viewing it as composed of only protons and electrons. It was known that atomic nuclei usually had about half as many positive charges as protons. This was often explained by proposing that nuclei also contained some "nuclear electrons" to neutralize the excess charge. Thus, the nitrogen -14 nucleus would be composed of 14 protons and 7 electrons to give it a charge of +7 but a mass of 14 atomic mass units.
In 1930, Bothe and Becker in Germany observed that when beryllium, boron, or lithium were bombarded by fast alpha particles, the bombarded substance emitted something of much greater penetrating power than the original alpha-particles. Experiments by Curie and Joliot in Paris confirmed the results, but experimenters were unable to explain them. Chadwick in England repeated the experiments and found that they could be explained if one made the assumption that uncharged particles of mass approximately equal to that of a proton were emitted from the nuclei of the bombarded material. He called the particles neutrons.
The emission of a neutron from a beryillium nucleus takes place according to the reaction
2He4 + 4Be9 → 6C12 + 0n1
where 0n1 is the symbol for a neutron.
The discovery of the neutron gave the first real insight into the structure of the nucleus of an atom. It explained why the atomic mass was usually around twice the number of protons.
Since neutrons have no charge, they produce no ionization on their passage through gases. In addition, they are not affected by an electric field. Thus, when they are used to bombard nuclei, they are not deflected by the electric field around a nucleus as are charged particles such as alpha particles.
Slow neutrons. Slow neutrons (i.e. low energy neutrons, energy less than one electron volt) can be obtained by passing fast neutrons through such substances as water, graphite, or blocks of paraffin. Collisions with nuclei slow them down. Slow neutrons can be detected by means of the alpha particles they eject from the nucleus of a boron atom, according to the reaction
0n1 + 5B10 → 3Li7 + 2He4
The ejected alpha particle then produces ionization which may be detected by a Geiger counter or an ionization chamber.
The element uranium. The element uranium exists in nature in three isotopic forms: U-238, U-235, and U-234. The most abundant form by far is U-238, which forms 99.3% of all naturally occurring uranium. The U-235 isotope forms about 0.7% . Only traces of U-234 have been found. These three isotopes all have the same chemical properties but have different nuclear properties.
When U-238 is bombarded with slow neutrons, these neutrons are captured by the U-238 nucleus. An unstable U-239 isotope is formed which emits a beta particle and forms a new radioactive element, neptunium, number 93. Neptunium is also unstable. It gives out a beta particle and forms another man-made element, plutonium, number 94. Neptunium and plutonium are termed trans-uranium elements since they have higher atomic numbers than uranium.
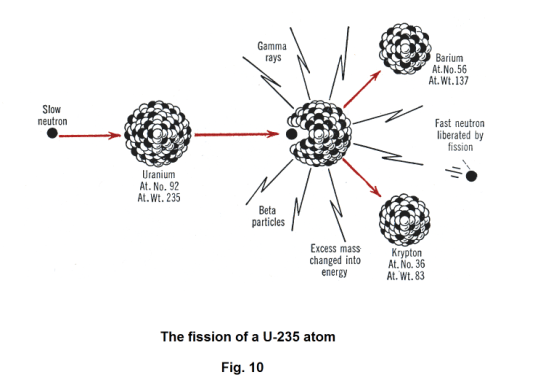
Nuclear fission. U-235 also captures slow neutrons when bombarded by them. The resulting nucleus is highly unstable. However, instead of emitting alpha or beta particles or gamma rays, the nucleus breaks up into two fairly equal parts. This break-up of the nucleus of an atom into two nuclei of more or less equal mass is called fission. During the fission neutrons are given out. See Fig. 10.
Nuclear fission was discovered by Hahn and Strassman in Germany in 1939. After bombarding uranium with neutrons, careful chemical analysis revealed barium and krypton among the products. Cloud-chamber photographs showed two heavy particles traveling in opposite directions with tremendous speed. Measurement showed an enormous amount of energy, 200 million electron volts, is released when uranium breaks up in this way.
Uranium fission can be accomplished using either fast or slow neutrons. U-238 and U-235 can both be split by a fast neutron. Only U-235 can be split by a slow neutron.
When uranium undergoes fission, barium and krypton are not the only products. Over one hundred different isotopes of more than 20 different elements have been detected among fission products.
Chain reactions. The discovery that neutrons are given out when uranium undergoes fission immediately raised the possibility of a chain reaction i.e. a process where one fission causes another fission which causes another fission in a self-sustaining chain of fissions. It also suggested the possibility of a chain reaction where one nuclear fission (releasing a huge amount of energy) immediately causes several more fissions, and each of those fissions immediately causes several more fissions, etc. in a mushrooming chain of fissions. Experimentation showed that a chain reaction was achievable and that it could be made to proceed slowly in a controlled manner. A controlled chain reaction is the underlying principle of the nuclear reactor. An uncontrolled chain reaction is the underlying principle of the atom bomb.
Thermonuclear reactions. There are two types of nuclear reactions in which large amounts of energy may be liberated. In both cases, the rest mass of the products are less than the rest mass of the reactants (where the loss in mass represents the amount of energy given off, as given by Einstein’s formula E = mc2). The fission of uranium is an example of one type. The other type involves two nuclei of low atomic weight combining to form a more complex nucleus of higher atomic weight in which the rest mass of the product is less than the rest mass of the reactants. This type of reaction is called fusion. Very high temperatures are needed to initiate a fusion reaction, temperatures of millions of degrees. However, an atom bomb achieves such temperatures and the hydrogen bomb, using an atomic bomb to initiate the reaction, employs the fusion process.
Uses of atomic energy. Nuclear reactors are used to generate electricity, produce power for atomic submarines, produce plutonium from U-238 for weapons, breed more fissionable material, and to produce radioactive isotopes for medical use and biological research.
Other elementary particles. Other elementary particles have been discovered. They include:
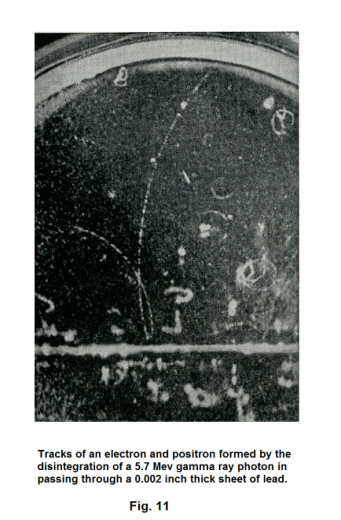
1. The positron. The positron is a particle of the same mass as the electron, but of positive sign. It was first observed by Dr. Carl D. Anderson in 1932 in an investigation of cosmic rays. They are ejected from the nuclei of certain artificially radioactive materials. They also spring into existence (along with a negative electron) in a process known as “pair production” in which a γ ray is simultaneously annihilated. See Fig. 11.
2. The meson. There are positive, negative and neutral mesons. Their masses are of the order of a few hundred electron masses. They were first observed by Anderson and Neddermeyer in 1936 as a component of cosmic radiation. The mass of a π-meson is about 280 times that of an electron. The mass of a μ-meson is about 210 times that of an electron. π-mesons are found in the fragments of nuclear explosions and have been produced in the laboratory by bombarding light nuclei with 400-Mev α particles. A π-meson is short lived , decaying in about 10-8 sec to a μ-meson.
Evolution in the concept of the structure of the atom. In the 1800's an atom was conceived of as being a simple mass (consisting of protons, etc.) in contrast to our present view of it as a tiny nucleus surrounded by shells of orbiting electrons at very large distances away. In our current view it is mostly empty space. The shift from the old view to the new view came as a direct consequence of an experiment performed by Ernest Rutherford in 1909 called the Rutherford gold foil experiment. The following is a Wikipedia account of that experiment:
Rutherford gold foil experiment
The discovery of the electron. The following articles give some background for the discovery of the electron.
J. J. Thomson is generally credited with being the discoverer of the electron. To understand why, read the following which contains a Nobel Lecture he gave in 1906.
Discovery of the Electron: J. J. Thomson
The discovery of the proton. As early as 1815, William Prout proposed that all atoms are composed of hydrogen atoms, based on early values of atomic weights.
In 1886, Eugen Goldstein discovered canal rays (also known as anode rays) and showed that they were positively charged particles (ions) produced from gases. However, since particles from different gases had different values of charge-to-mass ratio (e/m), they could not be identified with a single particle.
See Anode ray
Following the discovery of the atomic nucleus by Ernest Rutherford in 1911, Antonius van den Broek proposed that the place of each element in the periodic table (its atomic number) is equal to its nuclear charge. This was confirmed experimentally by Henry Moseley in 1913 using X-ray spectra.
In 1917, (in experiments reported in 1919) Rutherford proved that the hydrogen nucleus is present in other nuclei, a result usually described as the discovery of the proton. Rutherford had earlier learned to produce hydrogen nuclei as a type of radiation produced as a product of the impact of alpha particles on hydrogen gas, and recognize them by their unique penetration signature in air and their appearance in scintillation detectors. These experiments were begun when Rutherford had noticed that, when alpha particles were shot into air (mostly nitrogen), his scintillation detectors showed the signatures of typical hydrogen nuclei as a product. After experimentation Rutherford traced the reaction to the nitrogen in air, and found that when alphas were produced into pure nitrogen gas, the effect was larger. Rutherford determined that this hydrogen could have come only from the nitrogen, and therefore nitrogen must contain hydrogen nuclei. One hydrogen nucleus was being knocked off by the impact of the alpha particle, producing oxygen-17 in the process.
2He4 + 7N14 → 8O17 + 1H1
(This reaction would later be observed happening directly in a cloud chamber in 1925).
Rutherford knew hydrogen to be the simplest and lightest element and was influenced by Prout's hypothesis that hydrogen was the building block of all elements. Discovery that the hydrogen nucleus is present in all other nuclei as an elementary particle led Rutherford to give the hydrogen nucleus a special name as a particle, since he suspected that hydrogen, the lightest element, contained only one of these particles. He named this new fundamental building block of the nucleus the proton.
Excerpted from Wikipedia, “proton”
References
1. Dull, Metcalfe, Brooks. Modern Physics.
2. Sears, Zemansky. University Physics.
3. Semat, Katz. Physics.
4. Schaum. College Physics.
Jesus Christ and His Teachings
Way of enlightenment, wisdom, and understanding
America, a corrupt, depraved, shameless country
On integrity and the lack of it
The test of a person's Christianity is what he is
Ninety five percent of the problems that most people have come from personal foolishness
Liberalism, socialism and the modern welfare state
The desire to harm, a motivation for conduct
On Self-sufficient Country Living, Homesteading
Topically Arranged Proverbs, Precepts, Quotations. Common Sayings. Poor Richard's Almanac.
Theory on the Formation of Character
People are like radio tuners --- they pick out and listen to one wavelength and ignore the rest
Cause of Character Traits --- According to Aristotle
We are what we eat --- living under the discipline of a diet
Avoiding problems and trouble in life
Role of habit in formation of character
Personal attributes of the true Christian
What determines a person's character?
Love of God and love of virtue are closely united
Intellectual disparities among people and the power in good habits
Tools of Satan. Tactics and Tricks used by the Devil.
The Natural Way -- The Unnatural Way
Wisdom, Reason and Virtue are closely related
Knowledge is one thing, wisdom is another
My views on Christianity in America
The most important thing in life is understanding
We are all examples --- for good or for bad
Television --- spiritual poison
The Prime Mover that decides "What We Are"
Where do our outlooks, attitudes and values come from?
Sin is serious business. The punishment for it is real. Hell is real.
Self-imposed discipline and regimentation
Achieving happiness in life --- a matter of the right strategies
Self-control, self-restraint, self-discipline basic to so much in life